Services - Description of services
Computer System Validation
What is Computer System Validation? It’s the documented evidence that your system meets all regulatory and user requirements, defining the players involved and the specific responsibilities of a pharmaceutical business process managed by a computerized system.
Operating in the pharmaceutical environment, in fact, means following defined and regulated standard operating procedures, better known by the term SOPs (Standard Operating Procedures). The safeguard of the person and the care of the product quality are the starting point for the genesis of the production of a drug related standards. Validation, true to these principles, wants to formally document consistency and reliability not only of the production processes but also, in particular, equipment, systems, and services that have or may have a direct or potential impact on quality, purity and efficacy of medicines.
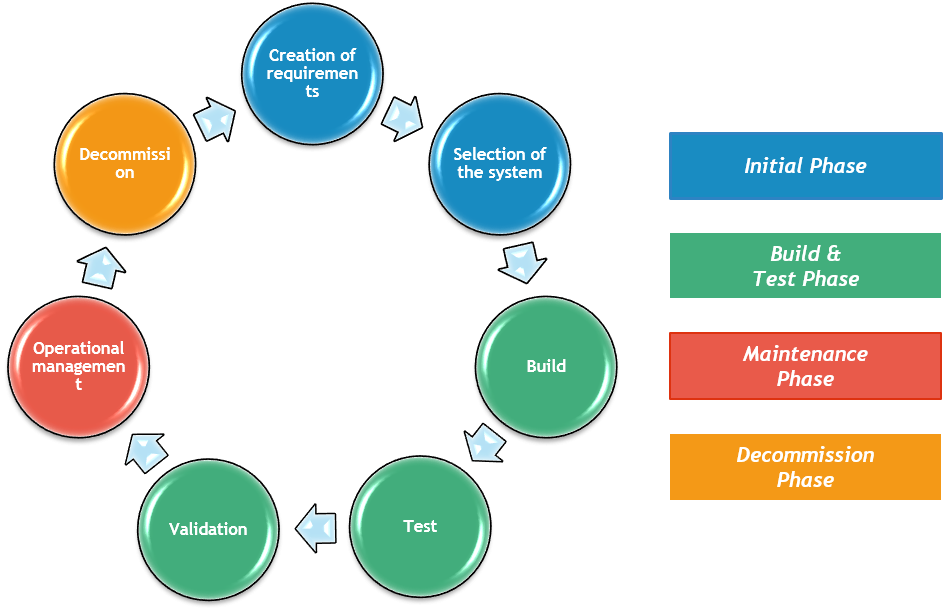
Computer System Validation represents the verification of the qualification of hardware and software based on the concept of Life Cycle and related phases: the initial phase (creation of requirements, selection of the system), the phase of Build and Test (build, testing, validation from the initial requirements), the maintenance phase (operations) and, finally, decommission.
Data Integrity
Data integrity is the degree to which the data are complete, consistent, accurate, reliable and trustworthy, and the way that all these features are maintained for the entire lifecycle of the data.
Data must be collected and kept in a secure way so that they can be attributable, legibly recorded in a contemporary way, either original or certified copies and accurate.
Ensuring Data Integrity requires appropriate quality systems for risk management, including also valid scientific principles and good documentation practices.
We provide a planned approach for the assessment, monitoring and management of data and the risks inherent to them, with the degree of management commensurate to the potential impact on health and patient safety and/or reliability of the decision made during all phases of the lifecycle of the data.
IT Compliance/QA
Regarding IT Compliance/QA, we support and provide:
- external audit conduction to the suppliers of IT systems and internals by national/international regulatory institutions to the customer
- QA Department of the customer for the assessment activities on the state of validation of computer systems, identifying remediation plans to the quality and compliance policies
- the creation and review of Standard Operating Procedures (for IT and/or operational management of the systems)
- feasibility studies for automation projects of processes and flows (ERP, LIMS, EDMS, MES, Data Warehouse, Document Management)
- assistance in the design and strategic choice (supplier selection) of infrastructure that meets the requirements of the regulated environments
